If the pressure of a gas sample is quadrupled and the absolute temperature is doubled by what factor does the volume of the. In fact if the volume increases by a certain factor the pressure decreases by the same factor and vice versa.
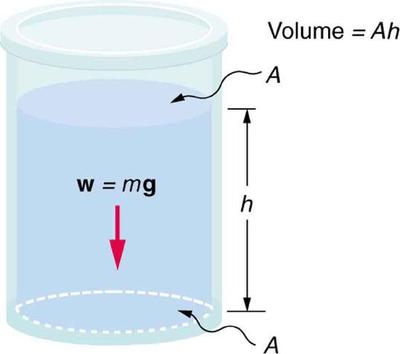
11 4 Variation Of Pressure With Depth In A Fluid Physics Libretexts

Someone Claims That The Absolute Pressure In A Liquid Of Constant Density Doubles When The Depth Is Brainly In

An Air Bubble Doubles In Radius On Rising From Bottom Of A Lake To Its Surface If The Atmosphere Youtube
Report as missing or 00 where the land fraction is 0.

Absolute pressure in liquid increase when depth is doubled. I p 11005100001013 993 atm. A pressure cooker version of PF Changs popular beef dish. Analysis No the absolute pressure in a liquid of constant density does not double when the depth is doubled.
An unusual property of water is that its solid formice frozen at atmospheric pressureis approximately 83 less dense than its liquid form. Decreasing the volume of a contained gas will increase its pressure and increasing its volume will decrease its pressure. Dispense the liquid into the receiving vessel by gently pressing the operating button.
For instance at 42 mm Hg pressure about 6 percent of atmospheric pressure Normal atmospheric pressure is 760 mm Hg millimeters of mercury column equivalent to 30 inches of Hg or 147 psi. Dip the tip into the solution to a depth of 1 cm and slowly release the operating button. It is the gage pressure that doubles when the depth is doubled.
At sea level the pressure of the air that surrounds us equal to 1 bar which is sometimes referred to as 1 atmosphere. If the submarine descends to a depth where the pressure is 150 atm a. 2-26 fluid 2 is carbon tetrachloride and fluid 1 is benzene.
Determine the gage pressure in the same liquid at a depth of 9 m. Volume-pressure data for an air sample at room temperature are graphed in Figure 5. Although the effect of thermal impact on the pore structure of shale gradually weakened as the thermal impact time increased it was clear that multiple thermal impacts have a more significant effect on the pore structure of shale compared with 13 thermal impacts.
Around 30 after 8 h of treatment. 7 Moderately elevated serum glucose levels may decrease coronary blood flow and cause myocyte death through either apoptosis or cellular injury resulting from exaggerated ischemic reperfusion. This Instant Pot Pressure Cooker Mongolian Beef is made with flank steak thinly sliced then cooked in a lightly sweet garlic ginger sauce until its melt in your mouth tender.
If the gas is an ideal gas what pressure would develop in the can if it were heated to 475C. Volume-pressure data for an air sample at room temperature are graphed in. 8-10 In acute hyperglycemia blood pressure.
The law has a simple mathematical form if the temperature is measured on an absolute scale such as in kelvins. As a function of longitude and latitude report the maximum soil depth reachable by plant roots ie the maximum soil depth from which they can extract moisture if defined in model. Withdraw the tip from the liquid touching it against the edge of the reservoir to remove excess liquid.
The cumulative increase in shale porosity has doubled from 353 in the first time to 735 in the seventh one. It is the gage pressure that doubles when the depth is doubled. In fact if the volume increases by a certain factor the pressure decreases by the same factor and vice versa.
Analysis No the absolute pressure in a liquid of constant density does not double when the depth is doubled. The pressure of 24 L of nitrogen gas in a flexible container is decreased to one-half its original pressure and its absolute temperature is increased to double the original temperature. A one-inch by one-inch column of water standing one foot tall will have a pressure at its base of 0434 pounds.
Academiaedu is a platform for academics to share research papers. Thus 6 percent of 147 psi is approximately 088 psi the temperature at the bottom of the column need only be about 35 C 95 F and the top about 20 C 68 F. Get 144 exercise solution 145 The absolute pressure in water at a depth of 5 m is read to be 145 kPa.
The gas pressure in an aerosol can is 18 atm at 25C. Discussion This is analogous to temperature scales when performing analysis using something like the ideal gas law you must use absolute temperature K not relative. Diabetes has doubled in New Zealand from 125000 to 250000 over the past 10 years where 90 225000 of these new diagnoses are T2DM Diabetes New Zealand 2019.
In fact the iron leaching was very similar at 0 1 and 2 bar. Pressure and temperature will both increase or decrease simultaneously as long as the volume is held constant. A small research submarine with a volume of 12 x 105 L has an internal pressure of 10 atm and an internal temperature of 150C.
Working Filters are placed in a vessel Slurry is pumped under pressure or occasionally by applying reduced pressure to the outlet side Slurry passes through the channels formed on the edges between the rings The clear liquid rises up and collected from the outlet into receiver 57 For separation of fine particles a bed of suitable materials such kieselguhr is first built up. 242 Assuming sea water to have a constant specificweight of 1005 kNm3 what is the absolute pressure at a depth of lOkm. This is equivalent to a volumetric expansion of 9.
Kelly Just a Taste shared a picture of her Mongolian Beef recipe on Facebook and it looked. It is a colorless odorless tasteless non-toxic inert monatomic gas the first in the noble gas group in the periodic table. The students cool the gas to 273K in an ice bath recording the temperature and pressure at regular intervals during the process.
However acute hyperglycemia negatively affects mechanisms--such as ischemic preconditioning--that protect the heart against ischemic damage. However a further increase of the pressure to 2 bar did not report a major improvement. Simplified this means that if you increase the temperature of a gas the pressure rises proportionally.
The gas is initially at a temperature of 293K. The downward pressure of a liquid is directly proportional to its depth. The students are surprised to find that the pressure remains constant as the temperature decreases.
For every person diagnosed with T2DM there is usually a family member or caregiver who also lives with diabetes in a support role. Moreover the effect of pressure on the catalyst was analyzed concluding that the integrity of the alginate beads was not compromised by pressure. A thermometer and a pressure sensor are attached to the container.
Decreasing the volume of a contained gas will increase its pressure and increasing its volume will decrease its pressure. Its boiling and melting point are the lowest among all the elementsHelium is the second lightest and second most abundant element in the. If Pam is 1015 kPa determine the absolute pressure at.
The density of ice is 09167 1 09168 2 gcm 3 at 0 C and standard atmospheric pressure 101325 Pa whereas water has a density of 09998 1 0999863 2 gcm 3 at the same. Determine a the local atmospheric pressure and b the absolute pressure at a depth of 5 m in a. Dive to just 10 meters deep and that pressure is doubled because water is much denser than air it exerts a much greater compressive force on your body.
Sun is a chemical element with the symbol He and atomic number 2. 3-3C Solution We are to examine a claim about absolute pressure.

Density And Pressure Boundless Physics

Density And Pressure Boundless Physics

Boyle S Law Wikipedia
Solved Part 1 Mcq 15 Answer The Following Questions Chegg Com

If The Gauge Pressure Is Doubled The Abso Clutch Prep

Chapter 13 Fluids

3 At What Depth Below The Surface Of Water Will Pressure Be Equal To Twice The Atmospheric Pressure The Atmospheric Pressure Is 10 N Cm Density Of Water Is 103 Kg
The Pressure Acting On A Submarine Is 3 X 10 Pa At Physics

